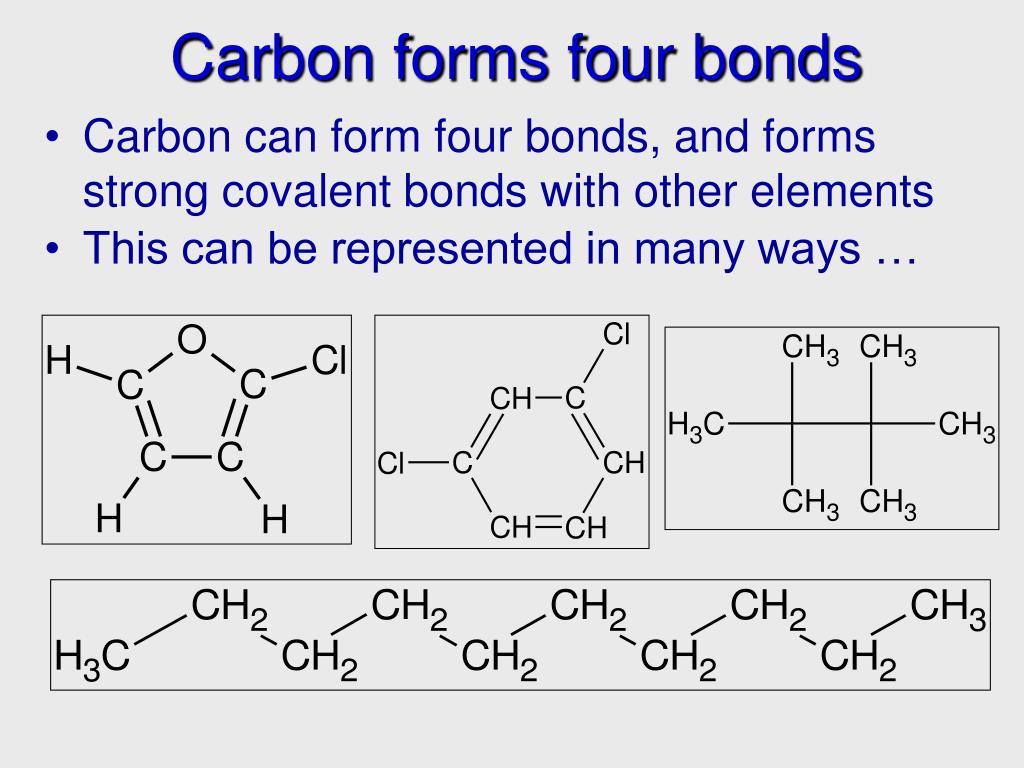
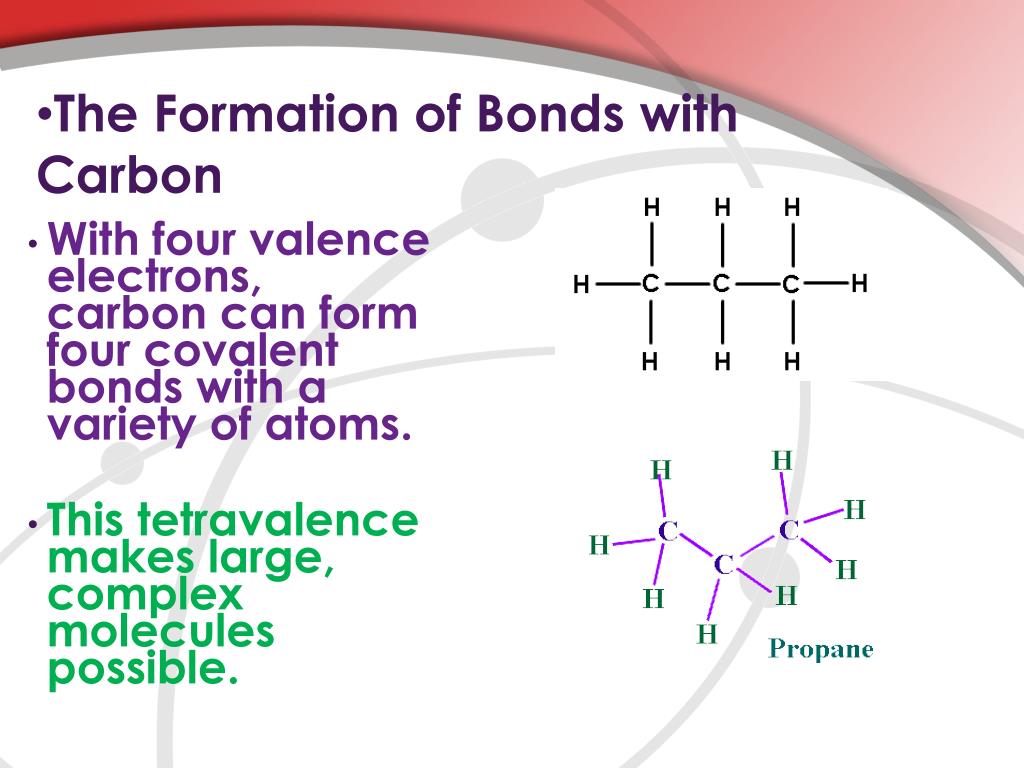
How Many Bonds Do Carbon Form - This allows carbon to fill its outer. Because carbon has four electrons in its valence (outer) shell, it can form four covalent bonds with other atoms or molecules. Carbon can form up to four covalent bonds and thus share four pairs of electrons with other atoms. How many bonds does carbon atom form. Carbon can form four covalent bonds to create an organic molecule. The simplest carbon molecule is methane (ch 4), depicted here. The correct answer and explanation is: A carbon atom can form four bonds.
The simplest carbon molecule is methane (ch 4), depicted here. Carbon can form four covalent bonds to create an organic molecule. Carbon can form up to four covalent bonds and thus share four pairs of electrons with other atoms. The correct answer and explanation is: A carbon atom can form four bonds. Because carbon has four electrons in its valence (outer) shell, it can form four covalent bonds with other atoms or molecules. This allows carbon to fill its outer. How many bonds does carbon atom form.
A carbon atom can form four bonds. How many bonds does carbon atom form. Carbon can form four covalent bonds to create an organic molecule. Carbon can form up to four covalent bonds and thus share four pairs of electrons with other atoms. Because carbon has four electrons in its valence (outer) shell, it can form four covalent bonds with other atoms or molecules. The simplest carbon molecule is methane (ch 4), depicted here. The correct answer and explanation is: This allows carbon to fill its outer.
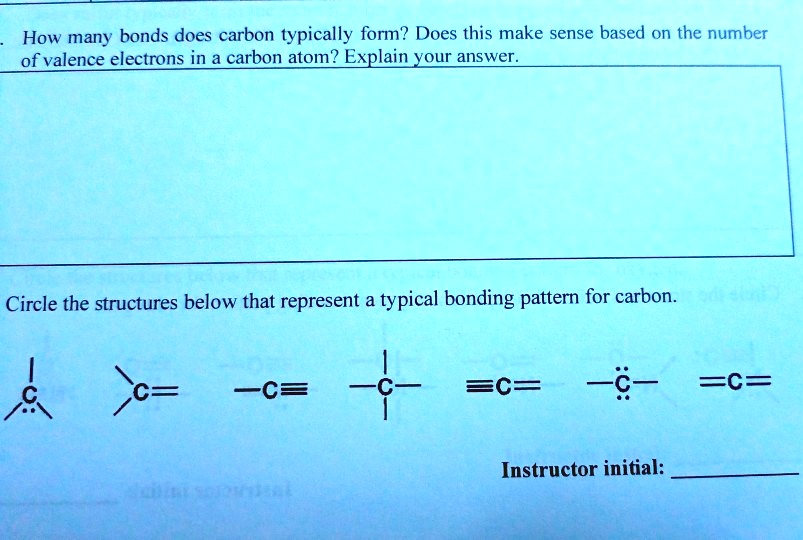
SOLVED How many bonds does carbon typically form? Does this make sense
Because carbon has four electrons in its valence (outer) shell, it can form four covalent bonds with other atoms or molecules. The correct answer and explanation is: A carbon atom can form four bonds. Carbon can form four covalent bonds to create an organic molecule. This allows carbon to fill its outer.
What Covalent Bonds Can Carbon Form at Douglas Jacobson blog
How many bonds does carbon atom form. A carbon atom can form four bonds. The correct answer and explanation is: This allows carbon to fill its outer. The simplest carbon molecule is methane (ch 4), depicted here.
MACROMOLECULES a.k.a. BioMolecules a.k.a. Organic Molecules ppt download
This allows carbon to fill its outer. The simplest carbon molecule is methane (ch 4), depicted here. The correct answer and explanation is: Carbon can form four covalent bonds to create an organic molecule. How many bonds does carbon atom form.
How To Determine The Number Of Bonds In A Molecule at Brock blog
The correct answer and explanation is: How many bonds does carbon atom form. Carbon can form up to four covalent bonds and thus share four pairs of electrons with other atoms. Carbon can form four covalent bonds to create an organic molecule. Because carbon has four electrons in its valence (outer) shell, it can form four covalent bonds with other.
[Class 10 Chemistry] Bonding in Carbon Atoms Covalent Bonds
A carbon atom can form four bonds. Carbon can form four covalent bonds to create an organic molecule. The simplest carbon molecule is methane (ch 4), depicted here. Carbon can form up to four covalent bonds and thus share four pairs of electrons with other atoms. Because carbon has four electrons in its valence (outer) shell, it can form four.
Carbon Dioxide Covalent Bond
The simplest carbon molecule is methane (ch 4), depicted here. How many bonds does carbon atom form. A carbon atom can form four bonds. The correct answer and explanation is: This allows carbon to fill its outer.
PPT Organic Chemistry Functional Groups PowerPoint Presentation
This allows carbon to fill its outer. The simplest carbon molecule is methane (ch 4), depicted here. Carbon can form four covalent bonds to create an organic molecule. Because carbon has four electrons in its valence (outer) shell, it can form four covalent bonds with other atoms or molecules. Carbon can form up to four covalent bonds and thus share.
How Many Forms Does Carbon Have at Claire Mcvicars blog
The simplest carbon molecule is methane (ch 4), depicted here. Carbon can form up to four covalent bonds and thus share four pairs of electrons with other atoms. Carbon can form four covalent bonds to create an organic molecule. This allows carbon to fill its outer. Because carbon has four electrons in its valence (outer) shell, it can form four.
Carbon Chemistry Carbon atoms can form single, double or triple bonds
Carbon can form up to four covalent bonds and thus share four pairs of electrons with other atoms. The correct answer and explanation is: Because carbon has four electrons in its valence (outer) shell, it can form four covalent bonds with other atoms or molecules. A carbon atom can form four bonds. Carbon can form four covalent bonds to create.
PPT Carbon PowerPoint Presentation, free download ID1833140
How many bonds does carbon atom form. The simplest carbon molecule is methane (ch 4), depicted here. Because carbon has four electrons in its valence (outer) shell, it can form four covalent bonds with other atoms or molecules. A carbon atom can form four bonds. Carbon can form four covalent bonds to create an organic molecule.
The Correct Answer And Explanation Is:
The simplest carbon molecule is methane (ch 4), depicted here. Carbon can form up to four covalent bonds and thus share four pairs of electrons with other atoms. How many bonds does carbon atom form. Because carbon has four electrons in its valence (outer) shell, it can form four covalent bonds with other atoms or molecules.
Carbon Can Form Four Covalent Bonds To Create An Organic Molecule.
A carbon atom can form four bonds. This allows carbon to fill its outer.
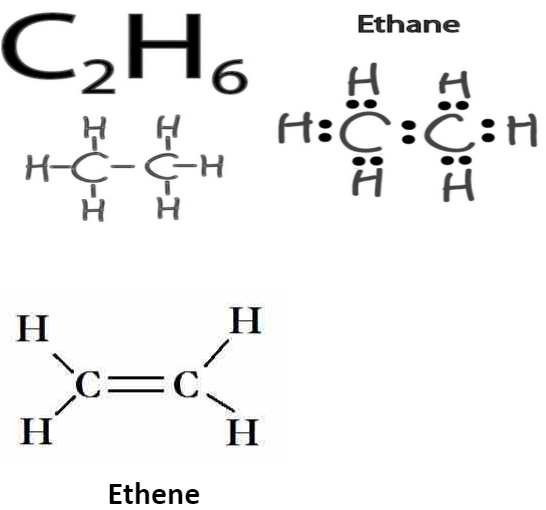


![[Class 10 Chemistry] Bonding in Carbon Atoms Covalent Bonds](https://d1avenlh0i1xmr.cloudfront.net/medium/693c5ecd-56f0-42a1-aba9-2e5a5be31300/covalent-bonding-in-co2---teachoo.jpg)