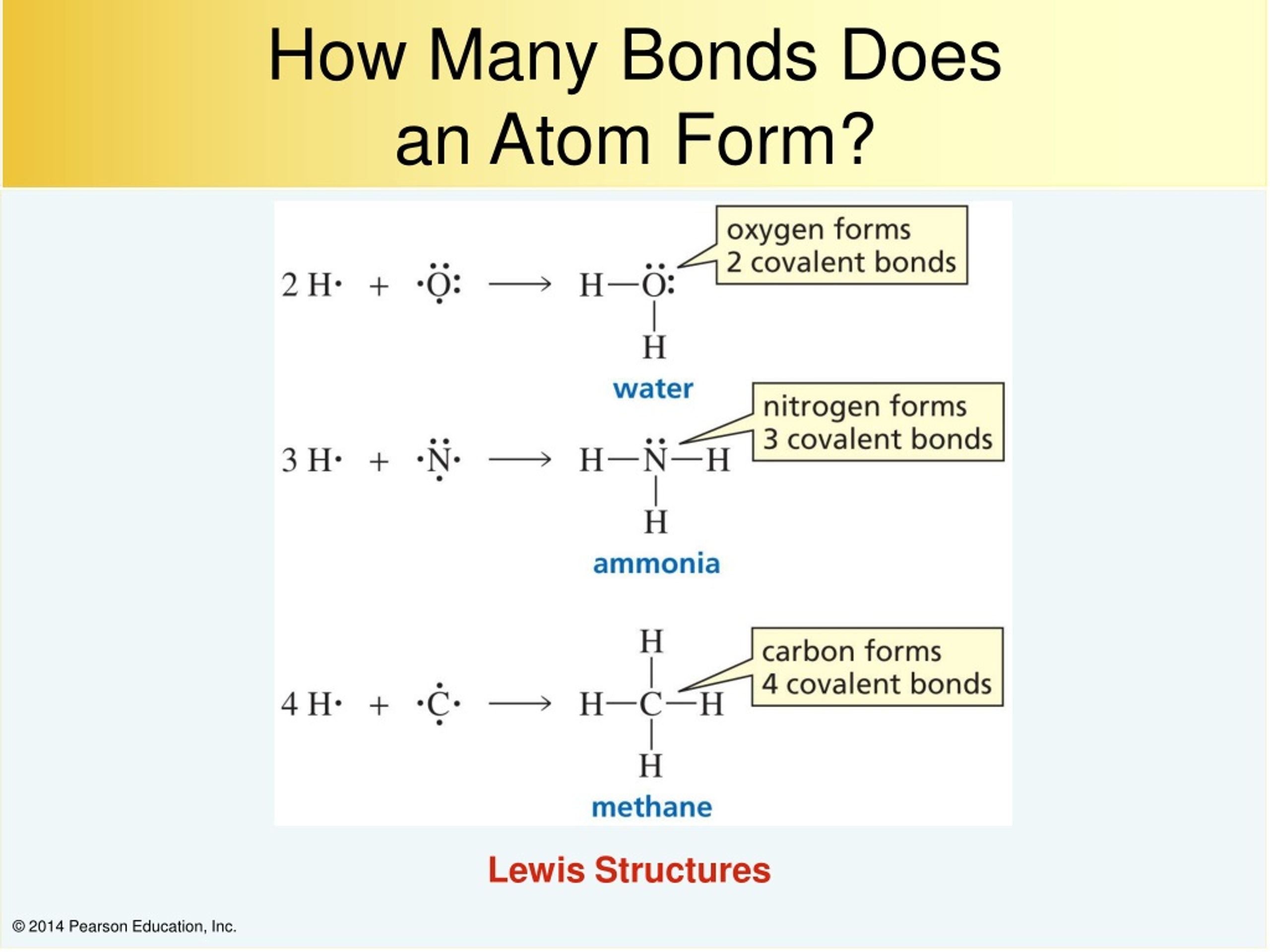
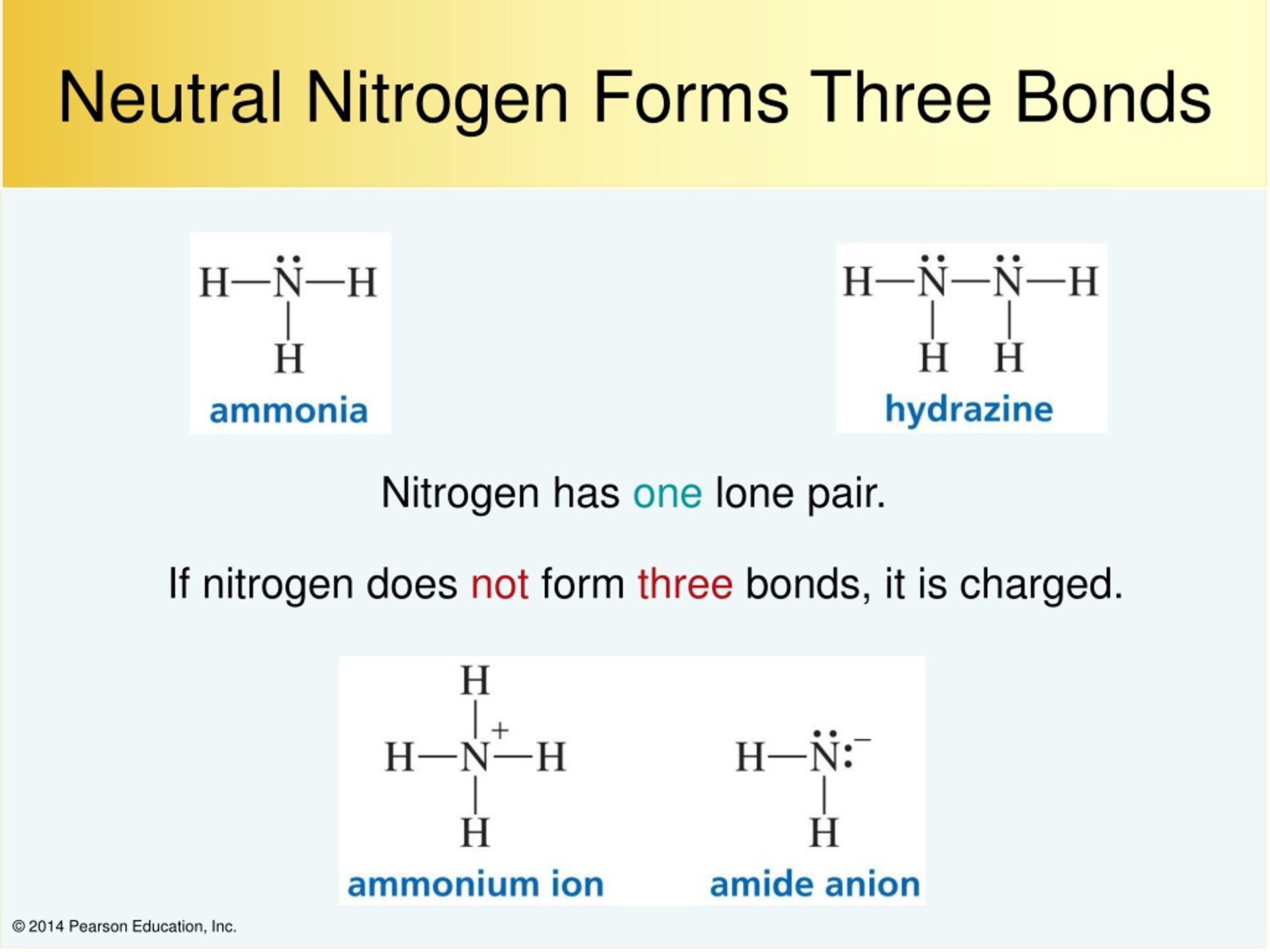
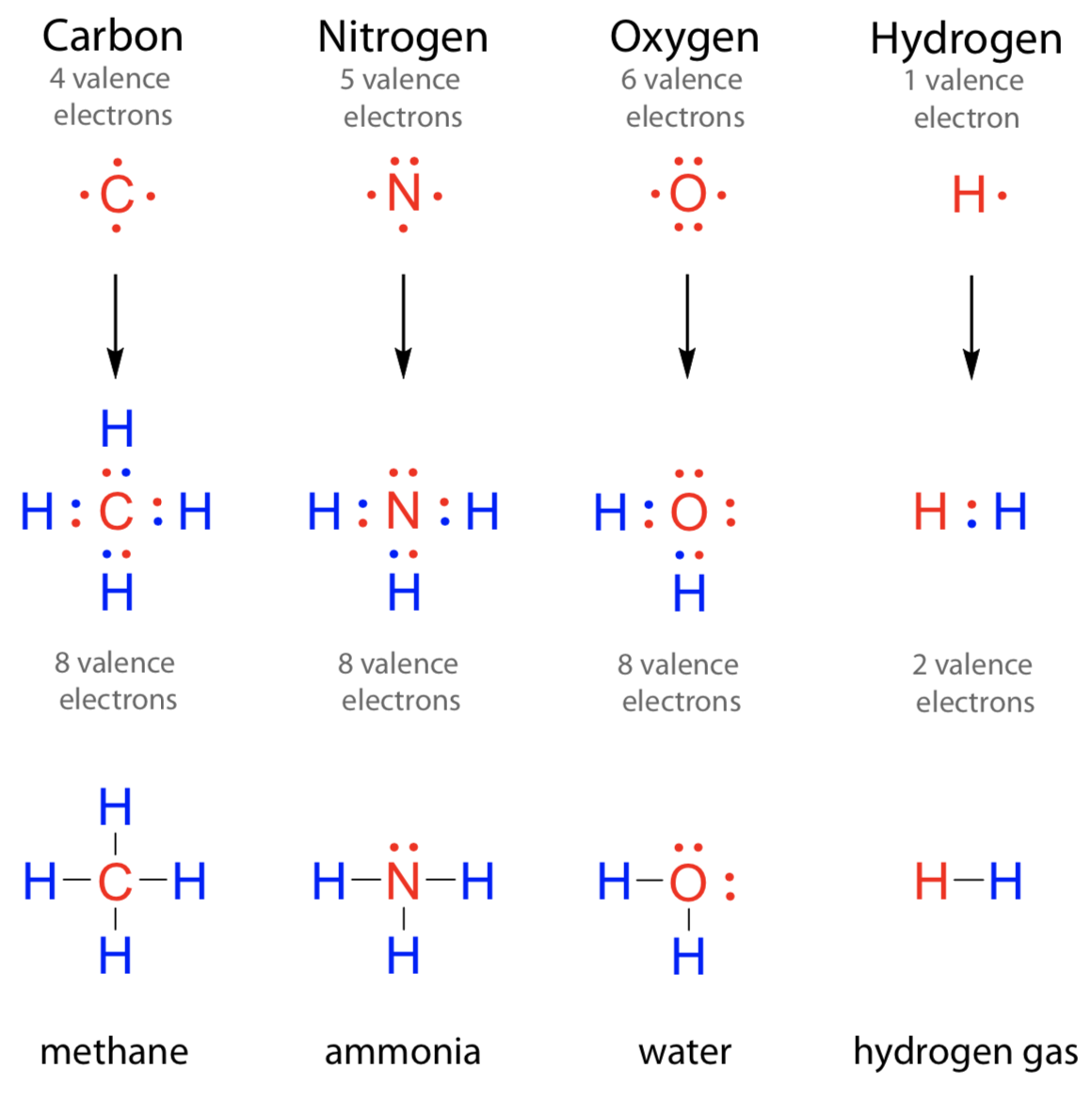
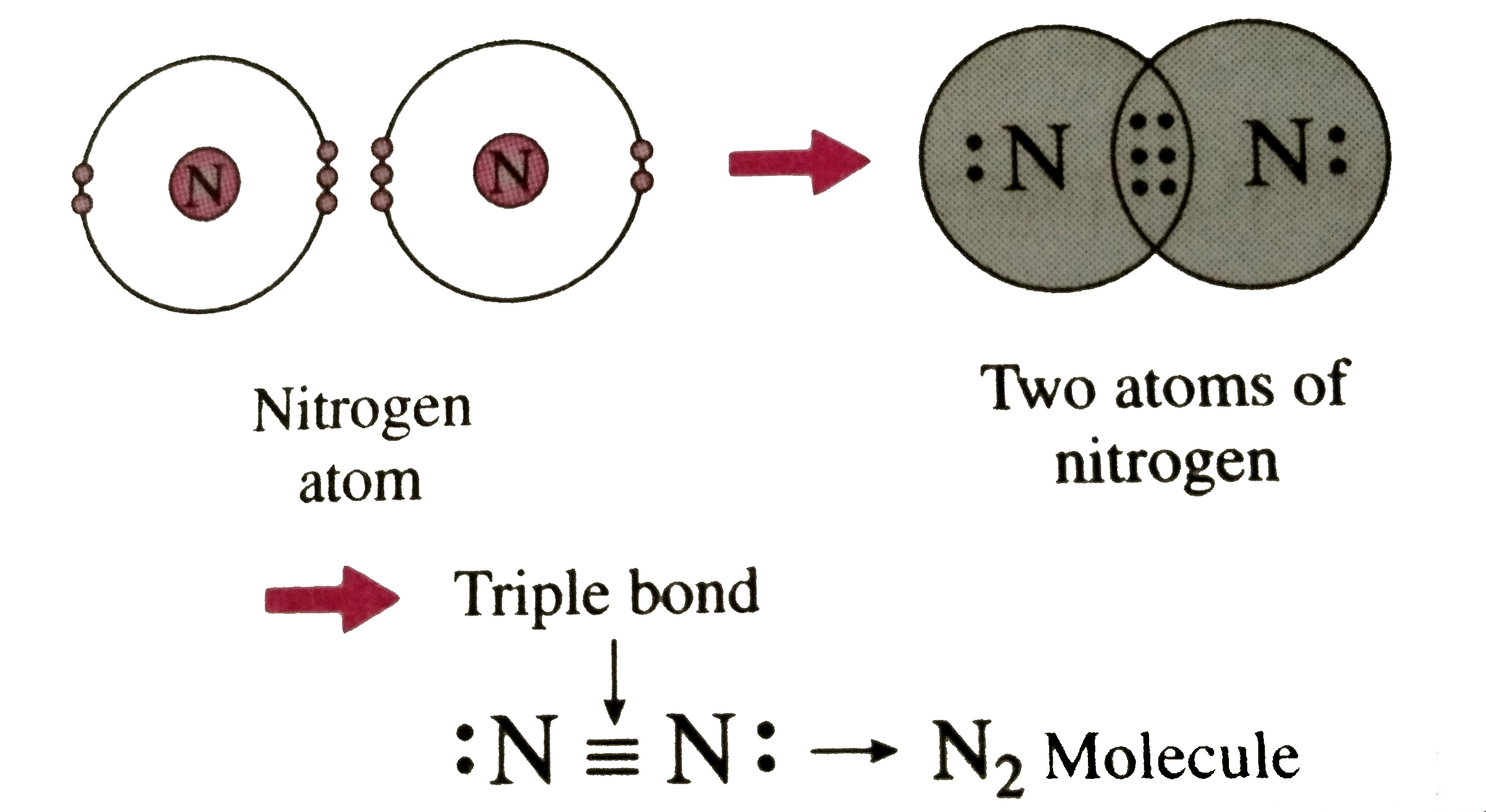How Many Bonds Does N Form - It is a noble gas and tends not to gain or lose electrons. However, with a positive net charge,. How many covalent bonds are in the element sb? An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration. Xenon (xe) typically forms covalent bonds. Hydrogen bonds form between a hydrogen atom bonded to an electronegative atom (such as oxygen, nitrogen, or fluorine) and another. In its pure form as an element, antimony (sb) is a metal, and it therefore forms a. K and cl would form an ionic bond as well.
K and cl would form an ionic bond as well. How many covalent bonds are in the element sb? An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration. Hydrogen bonds form between a hydrogen atom bonded to an electronegative atom (such as oxygen, nitrogen, or fluorine) and another. In its pure form as an element, antimony (sb) is a metal, and it therefore forms a. However, with a positive net charge,. It is a noble gas and tends not to gain or lose electrons. Xenon (xe) typically forms covalent bonds.
Hydrogen bonds form between a hydrogen atom bonded to an electronegative atom (such as oxygen, nitrogen, or fluorine) and another. An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration. K and cl would form an ionic bond as well. Xenon (xe) typically forms covalent bonds. However, with a positive net charge,. In its pure form as an element, antimony (sb) is a metal, and it therefore forms a. It is a noble gas and tends not to gain or lose electrons. How many covalent bonds are in the element sb?
PPT Remembering General Chemistry Electronic Structure and Bonding
In its pure form as an element, antimony (sb) is a metal, and it therefore forms a. It is a noble gas and tends not to gain or lose electrons. Xenon (xe) typically forms covalent bonds. However, with a positive net charge,. An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration.
PPT Remembering General Chemistry Electronic Structure and Bonding
K and cl would form an ionic bond as well. Xenon (xe) typically forms covalent bonds. How many covalent bonds are in the element sb? In its pure form as an element, antimony (sb) is a metal, and it therefore forms a. An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration.
Nitrogen Covalent Bond
It is a noble gas and tends not to gain or lose electrons. K and cl would form an ionic bond as well. Xenon (xe) typically forms covalent bonds. Hydrogen bonds form between a hydrogen atom bonded to an electronegative atom (such as oxygen, nitrogen, or fluorine) and another. In its pure form as an element, antimony (sb) is a.
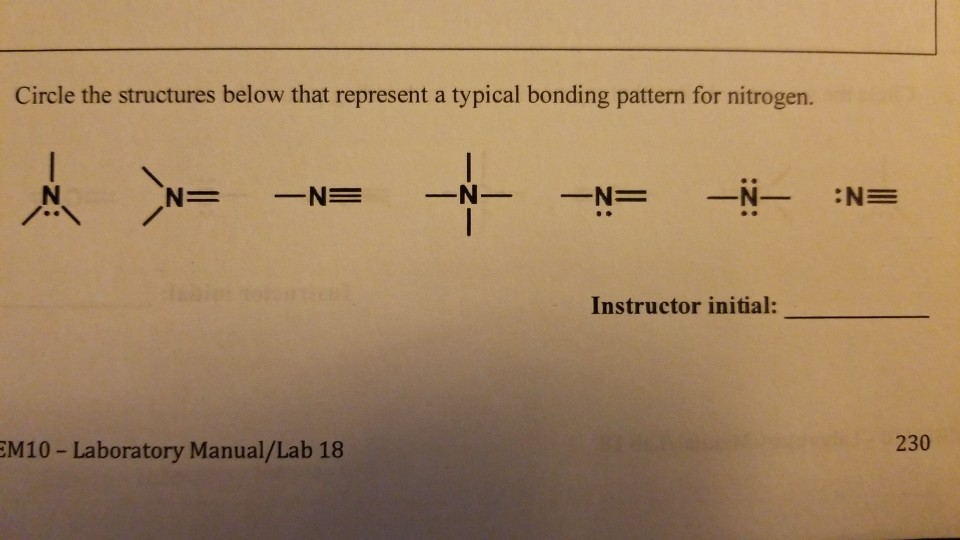
SOLVED How many bonds does nitrogen typically form? Does this make
Xenon (xe) typically forms covalent bonds. K and cl would form an ionic bond as well. An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration. Hydrogen bonds form between a hydrogen atom bonded to an electronegative atom (such as oxygen, nitrogen, or fluorine) and another. In its pure form as an element, antimony (sb).
Describe the formation of nitrogen molecule.
An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration. In its pure form as an element, antimony (sb) is a metal, and it therefore forms a. How many covalent bonds are in the element sb? It is a noble gas and tends not to gain or lose electrons. Hydrogen bonds form between a hydrogen.
Bond formation in nitrogen molecule Stock Image C028/6484 Science
Xenon (xe) typically forms covalent bonds. In its pure form as an element, antimony (sb) is a metal, and it therefore forms a. However, with a positive net charge,. An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration. How many covalent bonds are in the element sb?
Covalent Bond N2
K and cl would form an ionic bond as well. Hydrogen bonds form between a hydrogen atom bonded to an electronegative atom (such as oxygen, nitrogen, or fluorine) and another. An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration. How many covalent bonds are in the element sb? Xenon (xe) typically forms covalent bonds.
Solved How many bonds does nitrogen typically form? Does
However, with a positive net charge,. How many covalent bonds are in the element sb? In its pure form as an element, antimony (sb) is a metal, and it therefore forms a. Hydrogen bonds form between a hydrogen atom bonded to an electronegative atom (such as oxygen, nitrogen, or fluorine) and another. Xenon (xe) typically forms covalent bonds.
Nitrogen Covalent Bond
How many covalent bonds are in the element sb? Xenon (xe) typically forms covalent bonds. However, with a positive net charge,. K and cl would form an ionic bond as well. An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration.
LabXchange
In its pure form as an element, antimony (sb) is a metal, and it therefore forms a. Xenon (xe) typically forms covalent bonds. However, with a positive net charge,. K and cl would form an ionic bond as well. An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration.
How Many Covalent Bonds Are In The Element Sb?
Hydrogen bonds form between a hydrogen atom bonded to an electronegative atom (such as oxygen, nitrogen, or fluorine) and another. K and cl would form an ionic bond as well. In its pure form as an element, antimony (sb) is a metal, and it therefore forms a. Xenon (xe) typically forms covalent bonds.
It Is A Noble Gas And Tends Not To Gain Or Lose Electrons.
An atom of nitrogen typically forms 3 covalent bonds to achieve a stable electron configuration. However, with a positive net charge,.