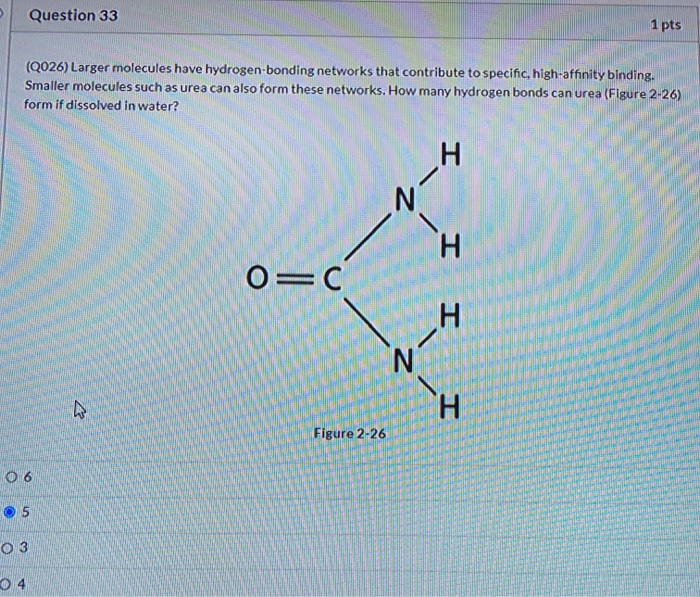
How Many Hydrogen Bonds Can Urea Form - The distinctive chemical characteristics of urea and its interactions with. Since there are two nitrogen atoms in urea, it can donate a total of 2 hydrogen bonds. Because of its chemical makeup, urea can generate five hydrogen bonds. Additionally, the carbonyl oxygen in urea can also. In urea, each nitrogen atom can form a hydrogen bond with a water molecule, and the oxygen atom can form two hydrogen bonds with two other. Smaller molecules such as urea can also form. Urea, in principle, can form 6 hydrogen bonds (involving both its carbonyl oxygen and the 4 amide hydrogens), but only the carbonyl oxygen.
Additionally, the carbonyl oxygen in urea can also. Smaller molecules such as urea can also form. The distinctive chemical characteristics of urea and its interactions with. Because of its chemical makeup, urea can generate five hydrogen bonds. In urea, each nitrogen atom can form a hydrogen bond with a water molecule, and the oxygen atom can form two hydrogen bonds with two other. Urea, in principle, can form 6 hydrogen bonds (involving both its carbonyl oxygen and the 4 amide hydrogens), but only the carbonyl oxygen. Since there are two nitrogen atoms in urea, it can donate a total of 2 hydrogen bonds.
Urea, in principle, can form 6 hydrogen bonds (involving both its carbonyl oxygen and the 4 amide hydrogens), but only the carbonyl oxygen. Since there are two nitrogen atoms in urea, it can donate a total of 2 hydrogen bonds. In urea, each nitrogen atom can form a hydrogen bond with a water molecule, and the oxygen atom can form two hydrogen bonds with two other. The distinctive chemical characteristics of urea and its interactions with. Smaller molecules such as urea can also form. Additionally, the carbonyl oxygen in urea can also. Because of its chemical makeup, urea can generate five hydrogen bonds.
Postulated hydrogen bonds formed between urea, ChCl and water, (a
Because of its chemical makeup, urea can generate five hydrogen bonds. In urea, each nitrogen atom can form a hydrogen bond with a water molecule, and the oxygen atom can form two hydrogen bonds with two other. Smaller molecules such as urea can also form. Since there are two nitrogen atoms in urea, it can donate a total of 2.
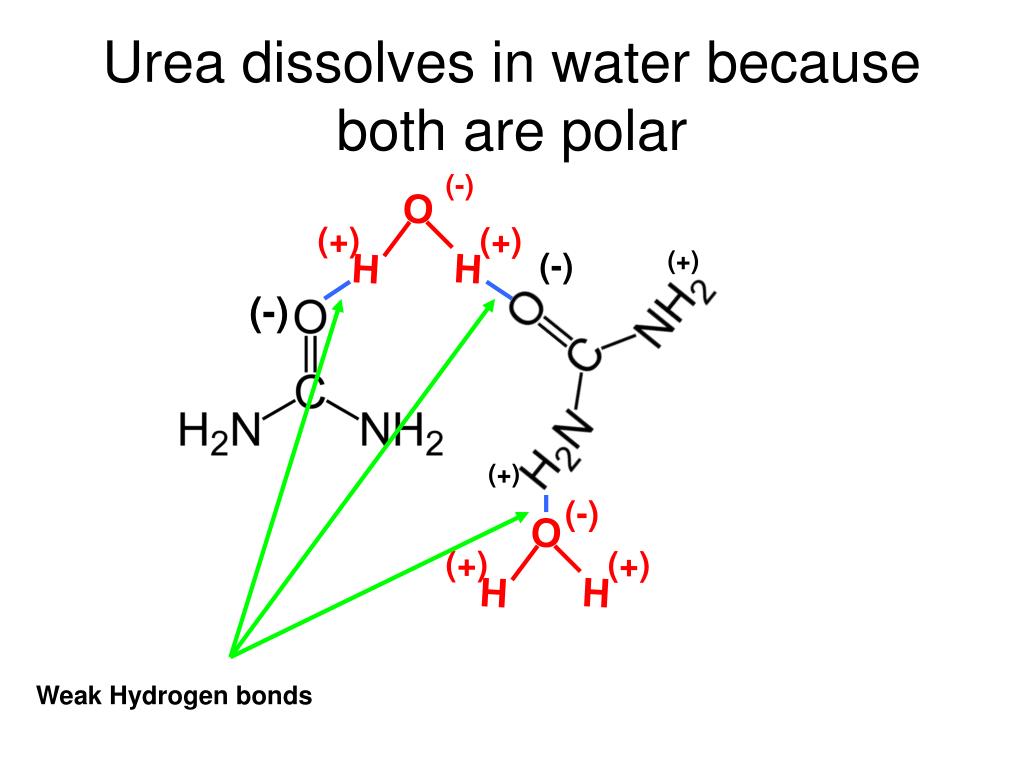
Urea is an organic compound widely used as a fertilizer. Its solubility
Urea, in principle, can form 6 hydrogen bonds (involving both its carbonyl oxygen and the 4 amide hydrogens), but only the carbonyl oxygen. Smaller molecules such as urea can also form. The distinctive chemical characteristics of urea and its interactions with. Since there are two nitrogen atoms in urea, it can donate a total of 2 hydrogen bonds. Additionally, the.
PPT Chapter 22 Properties of Water PowerPoint Presentation, free
Since there are two nitrogen atoms in urea, it can donate a total of 2 hydrogen bonds. Smaller molecules such as urea can also form. Urea, in principle, can form 6 hydrogen bonds (involving both its carbonyl oxygen and the 4 amide hydrogens), but only the carbonyl oxygen. In urea, each nitrogen atom can form a hydrogen bond with a.
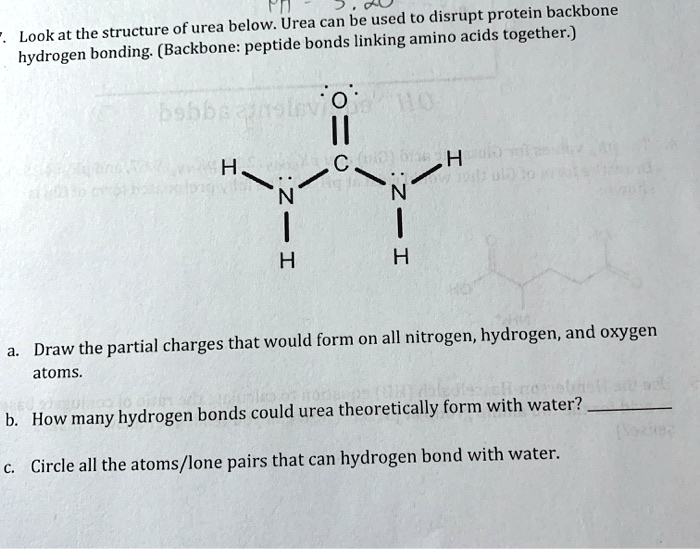
SOLVED Fn can be used to disrupt protein backbone. Look at the
Additionally, the carbonyl oxygen in urea can also. Since there are two nitrogen atoms in urea, it can donate a total of 2 hydrogen bonds. The distinctive chemical characteristics of urea and its interactions with. In urea, each nitrogen atom can form a hydrogen bond with a water molecule, and the oxygen atom can form two hydrogen bonds with two.
The Excretory System. ppt download
Because of its chemical makeup, urea can generate five hydrogen bonds. Urea, in principle, can form 6 hydrogen bonds (involving both its carbonyl oxygen and the 4 amide hydrogens), but only the carbonyl oxygen. Smaller molecules such as urea can also form. Since there are two nitrogen atoms in urea, it can donate a total of 2 hydrogen bonds. The.
How Many Hydrogen Bonds Can Urea Form
Smaller molecules such as urea can also form. Additionally, the carbonyl oxygen in urea can also. Because of its chemical makeup, urea can generate five hydrogen bonds. In urea, each nitrogen atom can form a hydrogen bond with a water molecule, and the oxygen atom can form two hydrogen bonds with two other. Urea, in principle, can form 6 hydrogen.
Hydrogen bonds during reverse osmosis filtration of urea molecules
Since there are two nitrogen atoms in urea, it can donate a total of 2 hydrogen bonds. Smaller molecules such as urea can also form. The distinctive chemical characteristics of urea and its interactions with. Urea, in principle, can form 6 hydrogen bonds (involving both its carbonyl oxygen and the 4 amide hydrogens), but only the carbonyl oxygen. Additionally, the.
[GET ANSWER] 8. Urea is a watersoluble product of nitrogen metabolism
The distinctive chemical characteristics of urea and its interactions with. Since there are two nitrogen atoms in urea, it can donate a total of 2 hydrogen bonds. Urea, in principle, can form 6 hydrogen bonds (involving both its carbonyl oxygen and the 4 amide hydrogens), but only the carbonyl oxygen. Because of its chemical makeup, urea can generate five hydrogen.
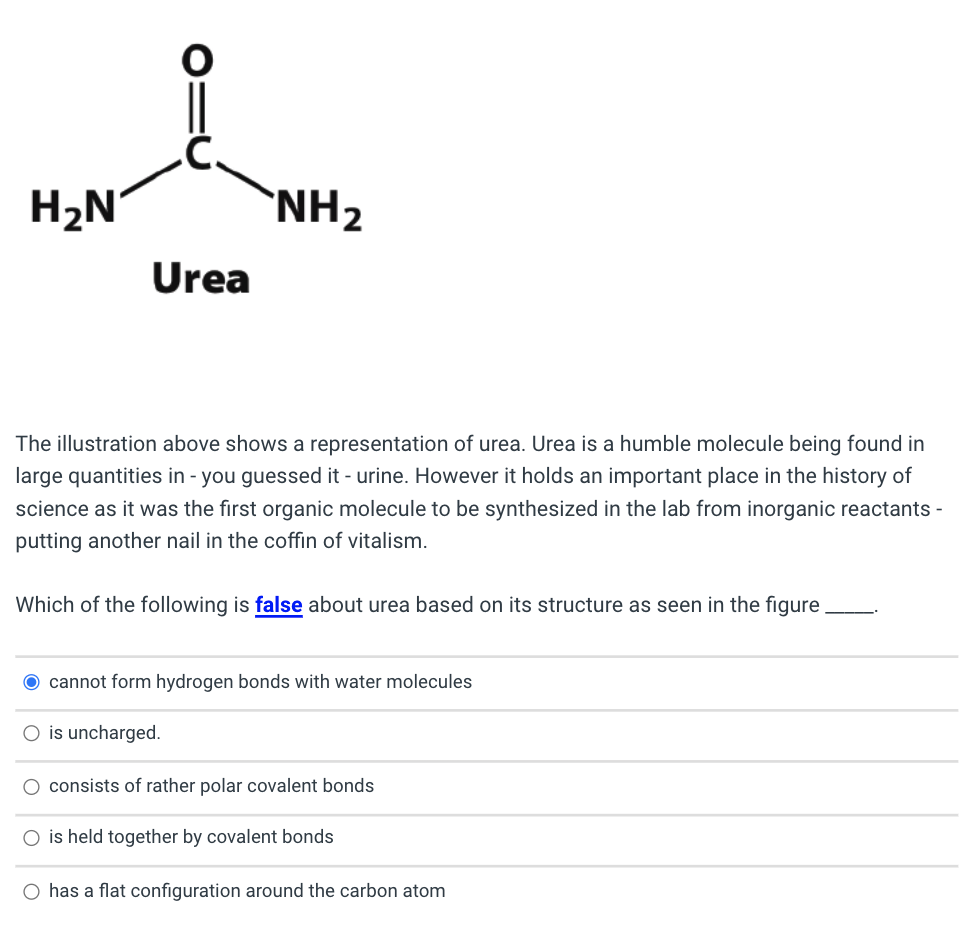
Solved H2N NH2 Urea The illustration above shows a
Since there are two nitrogen atoms in urea, it can donate a total of 2 hydrogen bonds. Additionally, the carbonyl oxygen in urea can also. Urea, in principle, can form 6 hydrogen bonds (involving both its carbonyl oxygen and the 4 amide hydrogens), but only the carbonyl oxygen. The distinctive chemical characteristics of urea and its interactions with. Smaller molecules.
(a) Hydrogen bonding around a urea molecule in the UC cocrystal. (b
Smaller molecules such as urea can also form. Because of its chemical makeup, urea can generate five hydrogen bonds. Since there are two nitrogen atoms in urea, it can donate a total of 2 hydrogen bonds. The distinctive chemical characteristics of urea and its interactions with. In urea, each nitrogen atom can form a hydrogen bond with a water molecule,.
Smaller Molecules Such As Urea Can Also Form.
The distinctive chemical characteristics of urea and its interactions with. Because of its chemical makeup, urea can generate five hydrogen bonds. Urea, in principle, can form 6 hydrogen bonds (involving both its carbonyl oxygen and the 4 amide hydrogens), but only the carbonyl oxygen. In urea, each nitrogen atom can form a hydrogen bond with a water molecule, and the oxygen atom can form two hydrogen bonds with two other.
Additionally, The Carbonyl Oxygen In Urea Can Also.
Since there are two nitrogen atoms in urea, it can donate a total of 2 hydrogen bonds.




![[GET ANSWER] 8. Urea is a watersoluble product of nitrogen metabolism](https://cdn.numerade.com/ask_images/6da2255547ef4048a23b3f1dd9991c8f.jpg)